Comparison of CH4 and CO2 Adsorptions onto Calcite(10.4), Aragonite(011)Ca, and Vaterite(010)CO3 Surfaces: An MD and DFT Investigation | ACS Omega

CaCO3(s) CaO(s) + CO₂(g) 3.25 mol CaCO3 decomposes according to the reaction above. What volume of CO2 gas - brainly.com
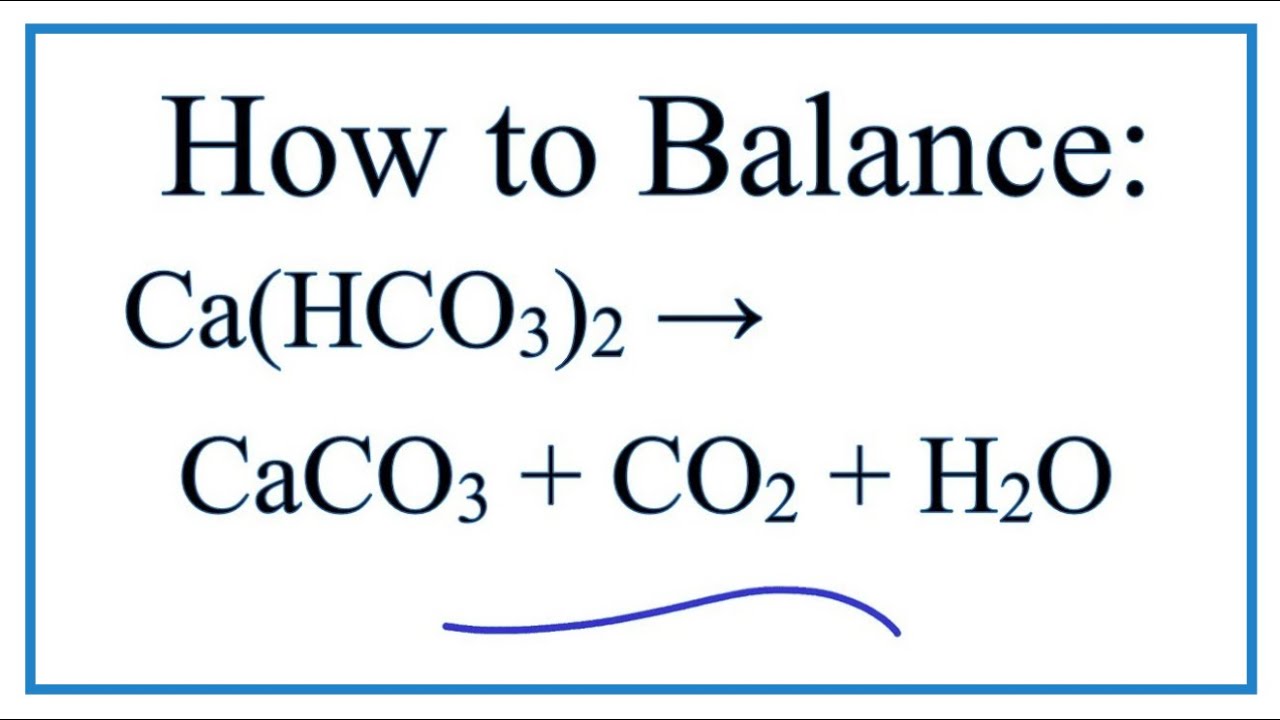
How to Balance Ca(HCO3)2 = CaCO3 + CO2 + H2O (Decomposition of Calcium hydrogen carbonate) - YouTube
CaO(s) + CO2(g) → CaCO3(s) + heat What is the total mass of CO2(s) needed to produce 300. grams of CaCO3(s)? - Quora
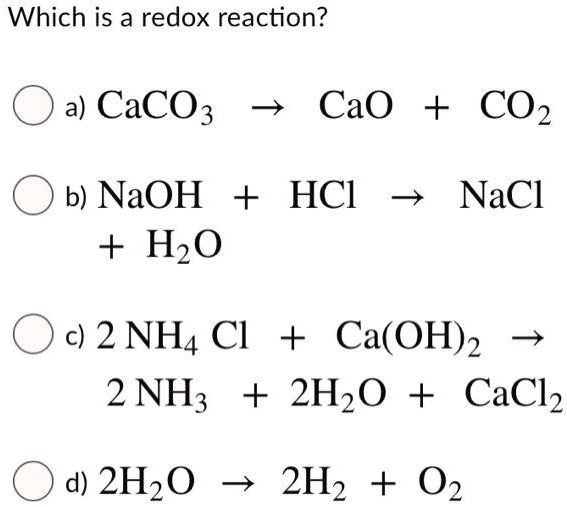
SOLVED: Which is a redox reaction? a) CaCO3 â†' CaO + CO2 b) NaOH + H2O â†' HCl + NaCl c) 2 NH4Cl + Ca(OH)2 â†' 2 NH3 + 2H2O + CaCl2 d) 2H2O â†' 2H2 + O2
![CaCO3→ CaO + CO2,Δ H = + 180KJ . If entropies of CaCO3,CaO and CO2 respectively are 93,39 and 213J/mol/K , then Δ S (total) [in J/K] at 300K is CaCO3→ CaO + CO2,Δ H = + 180KJ . If entropies of CaCO3,CaO and CO2 respectively are 93,39 and 213J/mol/K , then Δ S (total) [in J/K] at 300K is](https://dwes9vv9u0550.cloudfront.net/images/2234632/d247a5dd-b799-4377-806f-2628447e070f.jpg)
CaCO3→ CaO + CO2,Δ H = + 180KJ . If entropies of CaCO3,CaO and CO2 respectively are 93,39 and 213J/mol/K , then Δ S (total) [in J/K] at 300K is

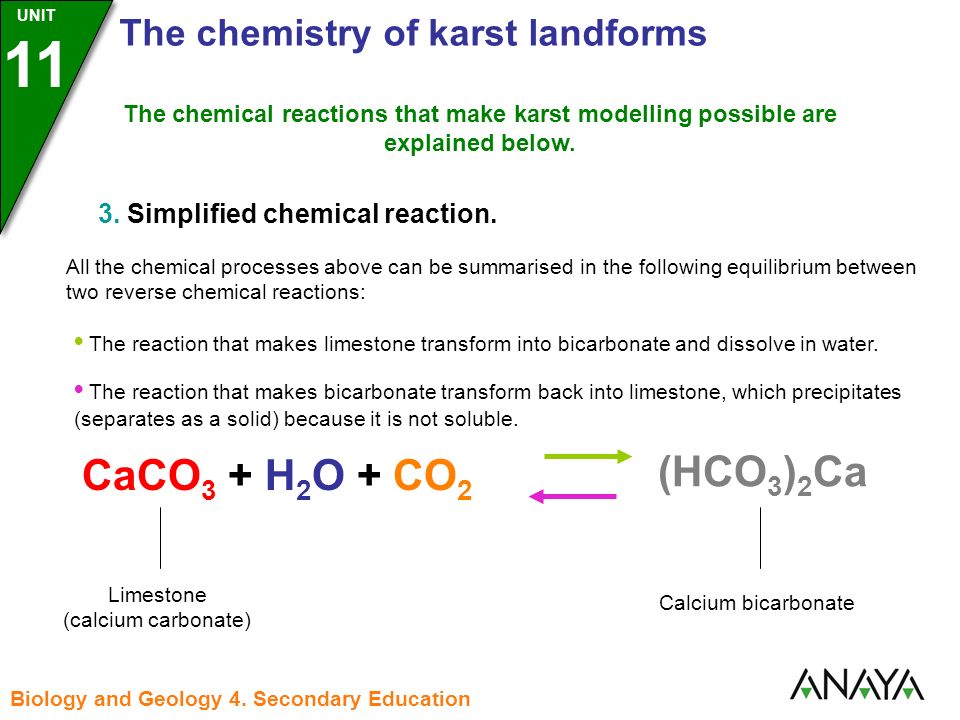
Summary of the reactions between carbon dioxide (CO2) with water (H2O)... | Download Scientific Diagram
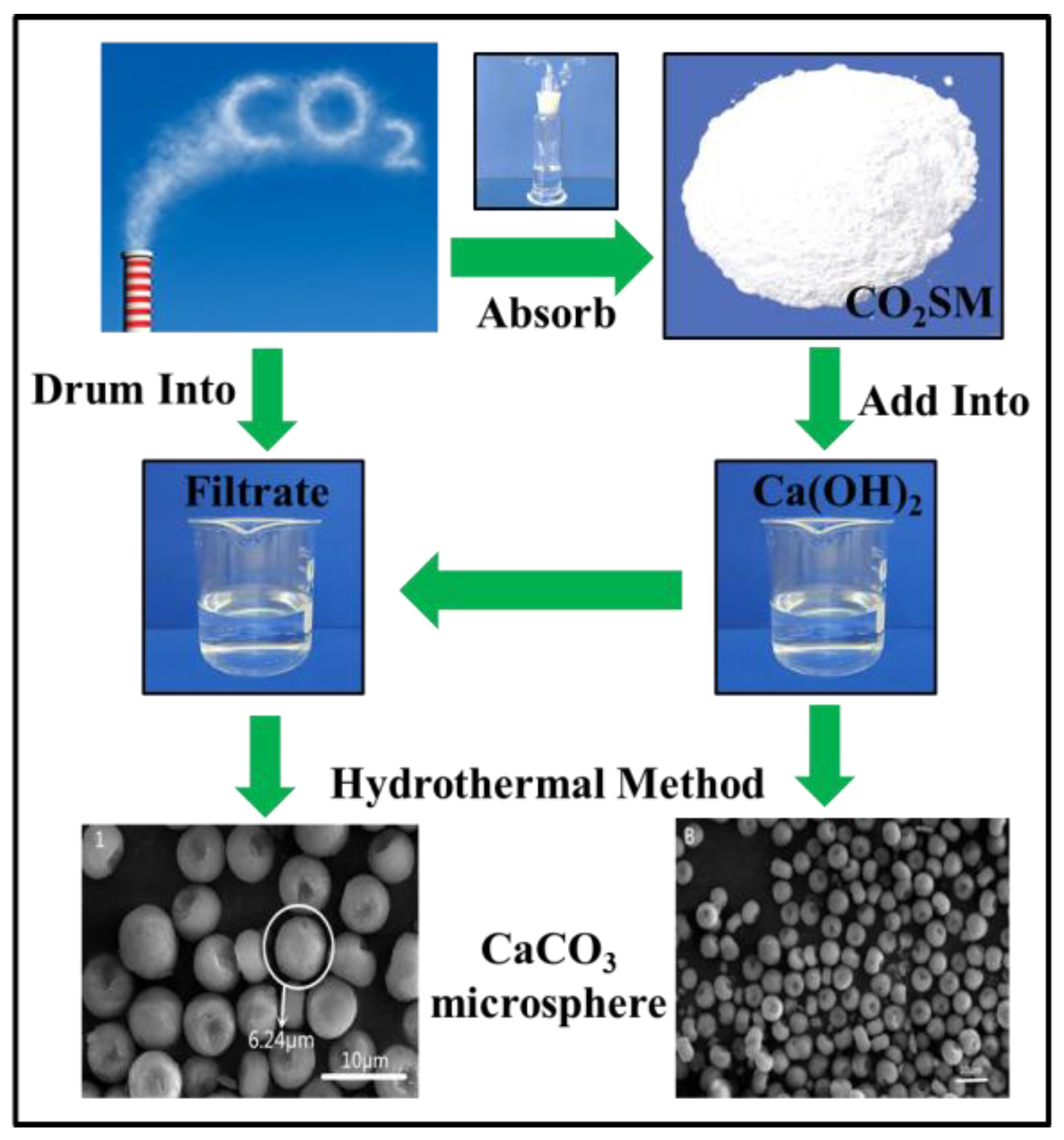
Crystals | Free Full-Text | Utilization of a CO2 Storage Material: Shape-Controlled Preparation of CaCO3 Microspheres
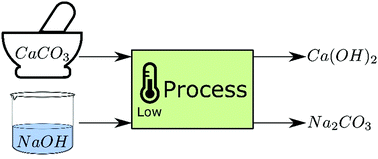
If 220 grams of calcium oxide (CaO) reacts with 50L of carbon dioxide (CO2), what mass of calcium carbonate (CaCO3) is produced? - Quora
![CaCO3→ CaO + CO2,Δ H = + 180KJ . If entropies of CaCO3,CaO and CO2 respectively are 93,39 and 213J/mol/K , then Δ S (total) [in J/K] at 300K is CaCO3→ CaO + CO2,Δ H = + 180KJ . If entropies of CaCO3,CaO and CO2 respectively are 93,39 and 213J/mol/K , then Δ S (total) [in J/K] at 300K is](https://dwes9vv9u0550.cloudfront.net/images/10263170/3e36f846-ca64-4ea9-be05-3cab519e4454.jpg)
CaCO3→ CaO + CO2,Δ H = + 180KJ . If entropies of CaCO3,CaO and CO2 respectively are 93,39 and 213J/mol/K , then Δ S (total) [in J/K] at 300K is
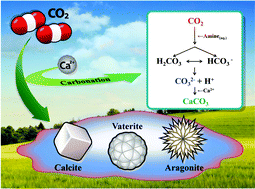
CO2 mineralization into different polymorphs of CaCO3 using an aqueous-CO2 system - RSC Advances (RSC Publishing)