Sum of purchase_quantity Months drugfull drug_packsize Jan Feb Mar Apr May Jun Jul 3 CHAMBER FK WITH ADDITIONS (3C-68) Intraven
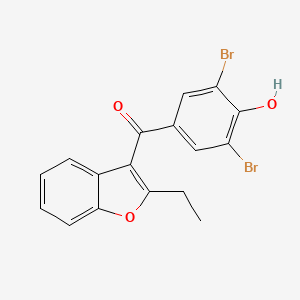
Type II variation Public Assessment Report Desuric, tablets 100 mg (benzbromarone) Marketing Authorisation Holder: ProStrakan L

PDF) A benefit-risk assessment of benzbromarone in the treatment of gout. Was its withdrawal from the market in the best interest of patients?