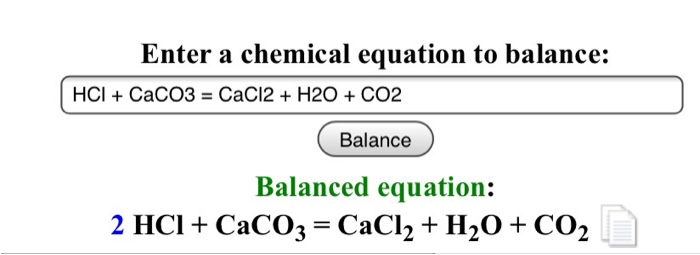
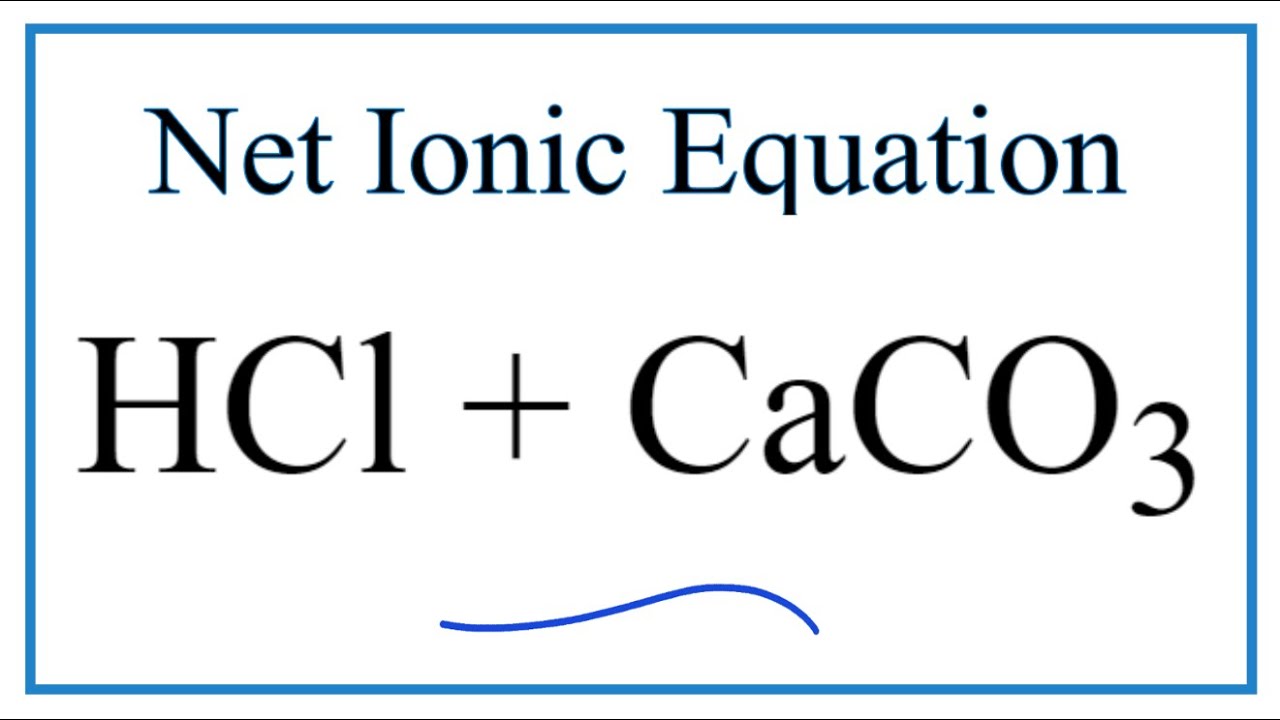16. In a chemical reaction, caco3+2hcl= cacl2 +co2+h2o. 25ml hcl and 0.75M Calculate the amount of caco3
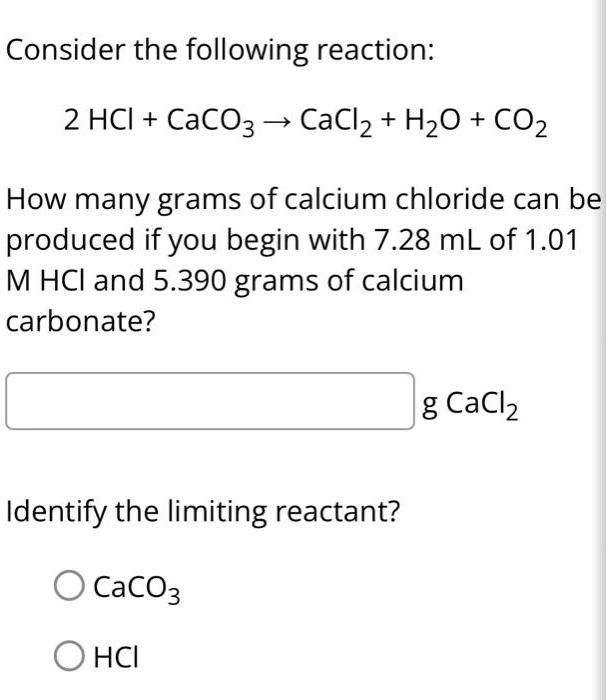
SOLVED: Consider the following reaction: 2 HCl + CaCO3 -> CaCl2 + H2O + CO2 How many grams of calcium chloride can be produced if you begin with 7.28 mL of 1.01
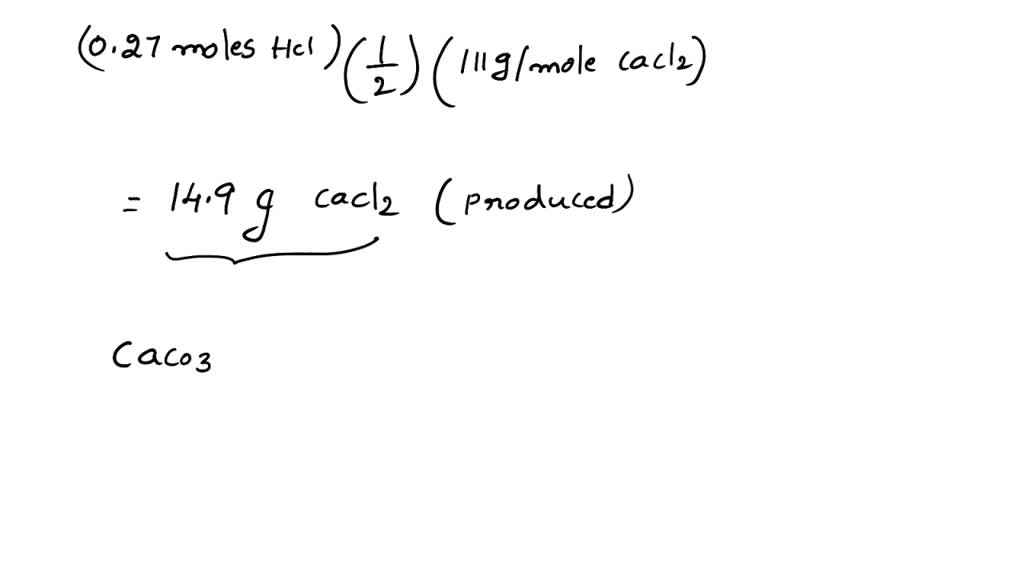
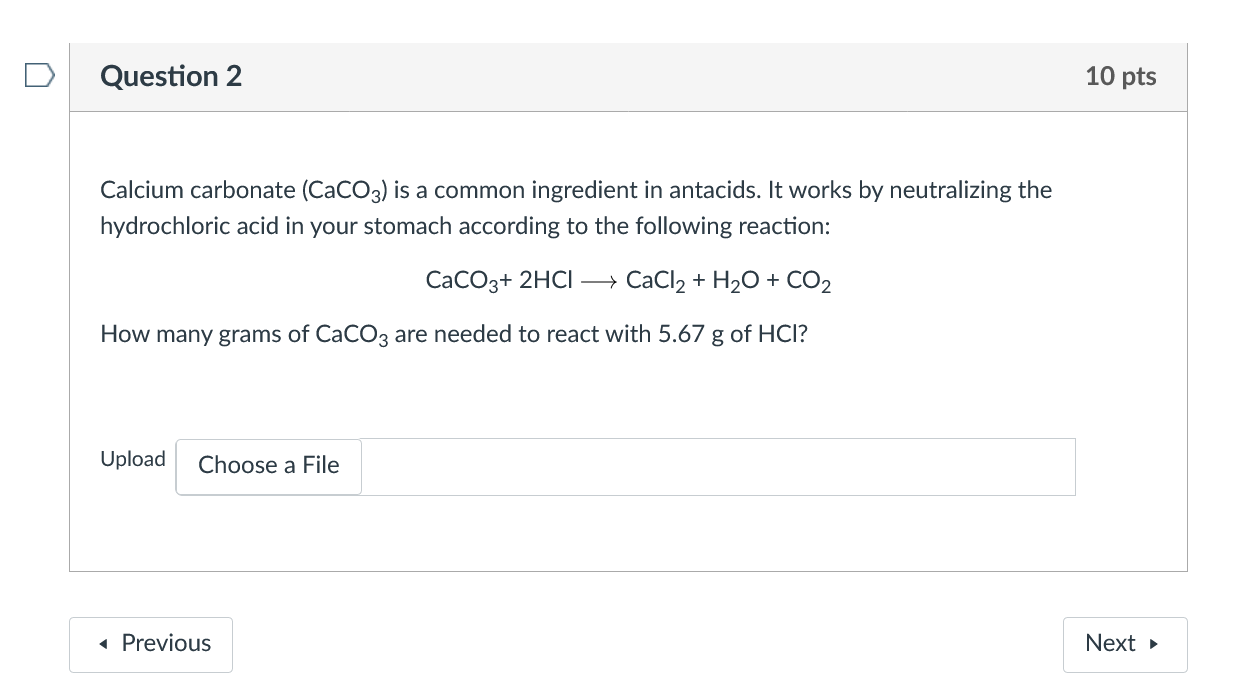
SOLVED: Calcium carbonate reacts with dilute hydrochloric acid. The equation for the reaction is shown: CaCO3 + 2HCl = CaCl2 + H2O + CO2. 1 g of calcium carbonate is added to

Calcium carbonate react with aqueous HCI to given below: CaCO3(s) + 2HCl (aq) → CaCl2 + CO2 + H2O What mass - Brainly.in
To prepare carbon dioxide gas, 25.0 of calcium carbonate is added to a solution in which 20g hydrogen chloride has been dissolved. The unbalanced reaction is CaCo3 +HCl=CaCl2 +CO2+H2O…determine the limiting reagent? -

Question Video: Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution | Nagwa
40. Consider the reaction CaCO3+2HCL (l) 》CaCl2+CO2+H2O (l).what mass of CaCO3 is required to react with 20mL 1M HCL?