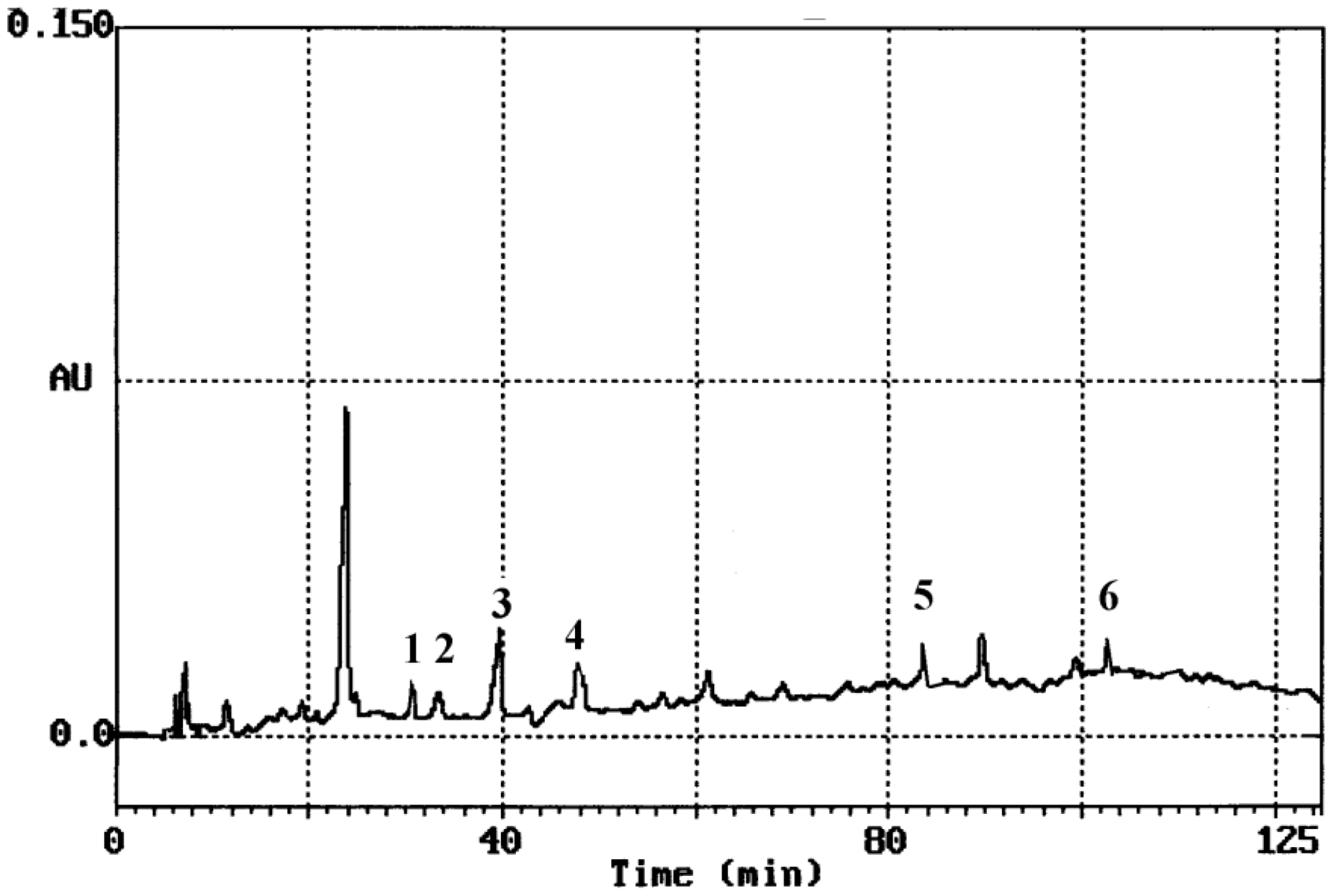
The use of silylation for minimizing the interference effects caused by contamination in ultra-low level mercury analytics
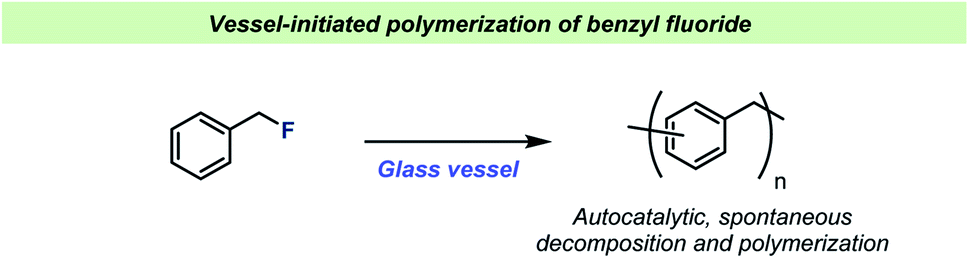
Vessel effects in organic chemical reactions; a century-old, overlooked phenomenon - Chemical Science (RSC Publishing) DOI:10.1039/D2SC01125E

Silylation of N-(2-hydroxyphenyl)acetamide by methyl(organyl)dichlorosilanes: Structure and properties of resulting heterocycles - ScienceDirect

Alkylation versus trans-silylation of N-methyl-N-trimethylsilylacetamide with ambident electrophiles (chloromethyl)fluorosilanes - ScienceDirect
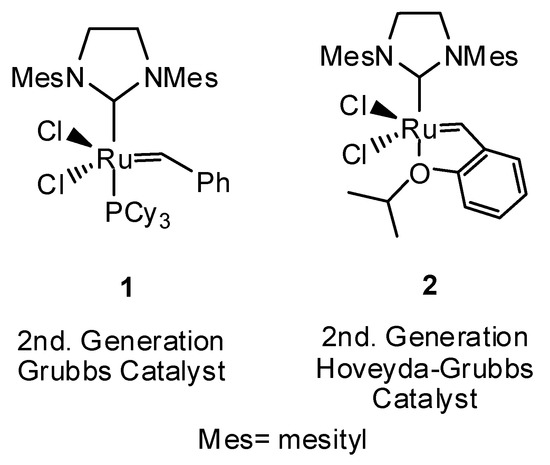
Catalysts | Free Full-Text | Unprecedented Multifunctionality of Grubbs and Hoveyda–Grubbs Catalysts: Competitive Isomerization, Hydrogenation, Silylation and Metathesis Occurring in Solution and on Solid Phase

PDF) Rapid silylation of a glass surface: choice of reagent and effect of experimental parameters on hydrophobicity | F. Deyhimi - Academia.edu

Transition Metal‐Free Catalytic C−H Silylation of Terminal Alkynes with bis(Trimethylsilyl)acetylene Initiated by KHMDS - Kuciński - 2022 - ChemCatChem - Wiley Online Library

Persilylation of ferrocene: the ultimate discipline in sterically overcrowded metal complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/D3DT01133J

Deoxygenative silylation of aromatic carbonyl compounds with HSiCl3 in the presence of quaternary phosphonium chloride: A facile route to arylmethyltrichlorosilane - ScienceDirect

Revisiting the Mg/TMSCl/Dipolar Solvent System for Dearomatic Silylation of Aryl Carbonyl Compounds: Substrate Scope, Transformations, and Mechanistic Studies | The Journal of Organic Chemistry
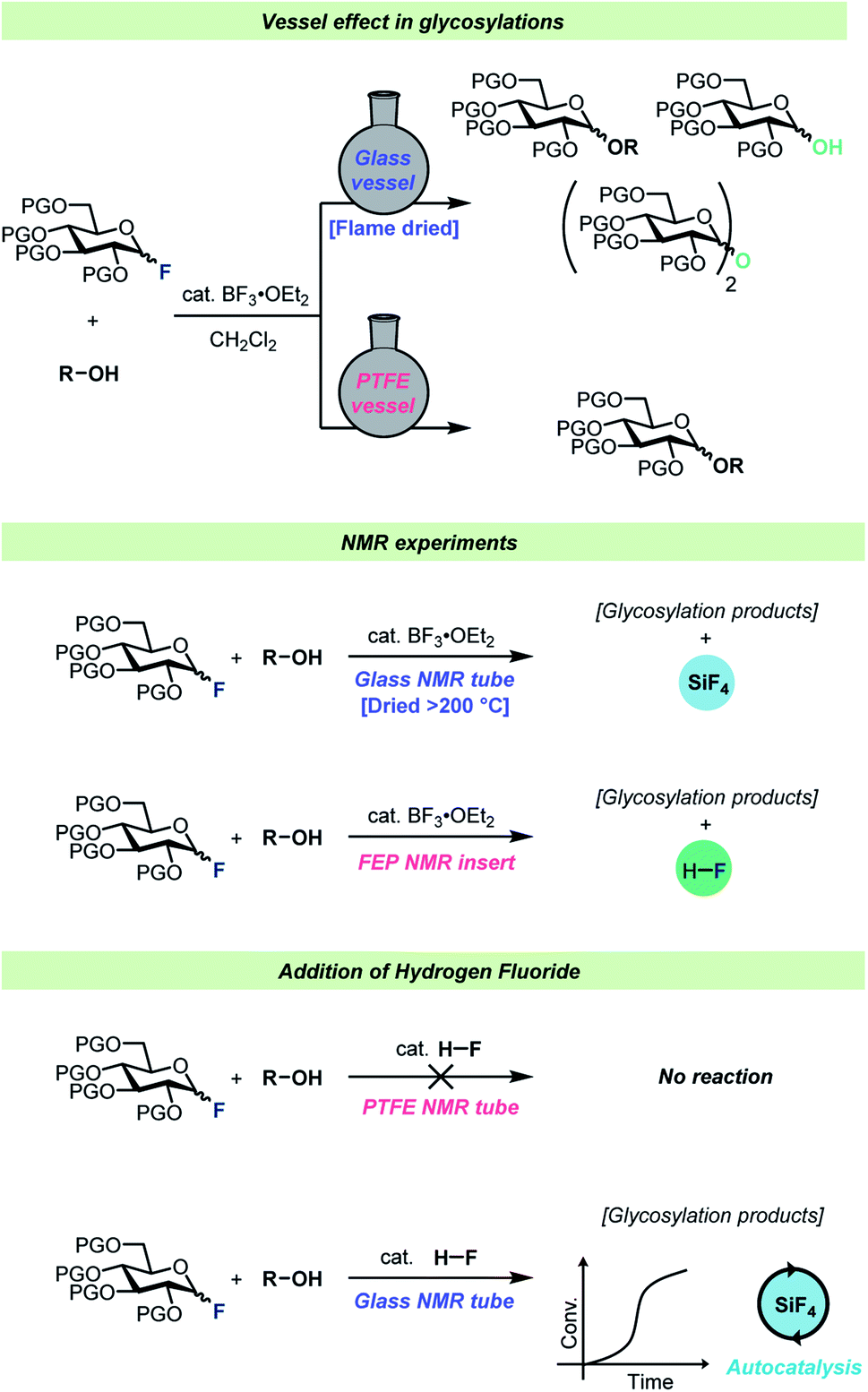
Vessel effects in organic chemical reactions; a century-old, overlooked phenomenon - Chemical Science (RSC Publishing) DOI:10.1039/D2SC01125E
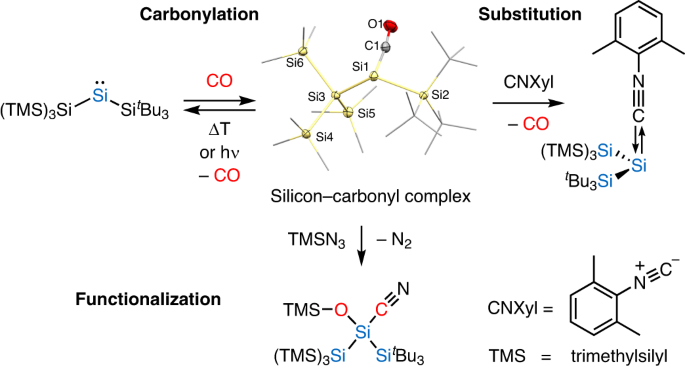
Silylated silicon–carbonyl complexes as mimics of ubiquitous transition-metal carbonyls | Nature Chemistry

Catalytic silylation of O–nucleophiles via Si–H or Si–C bond cleavage: A route to silyl ethers, silanols and siloxanes - ScienceDirect

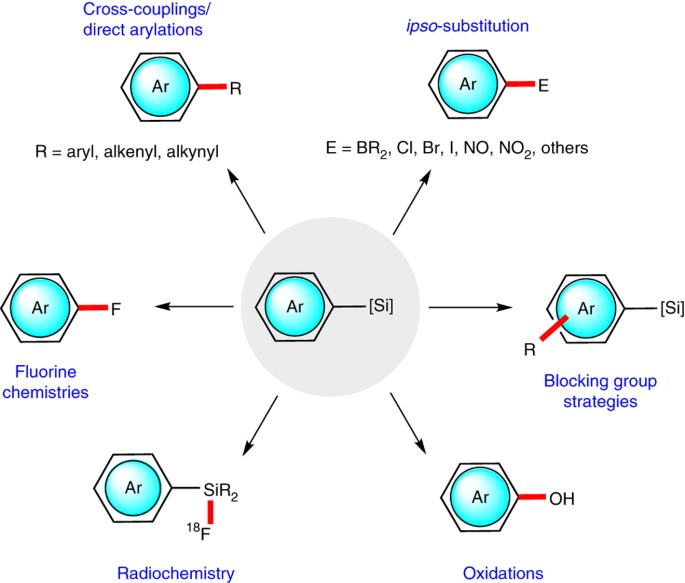
Scheme 16. Defluorinative silylation of substituted benzotrifluorides... | Download Scientific Diagram